The sources of the required organoboron compounds are manifold. The most popular organoboron compounds for this method, boronic acids and their esters (boronates), can be obtained through: (i) transmetalation from organolithium or organomagnesium compounds using B(OR) 3, or by transmetalation from other organometallics, e.g., stannanes 6 or silanes; 7–9. Boronic acids and esters are perhaps the most studied and most synthetically useful organoboron compounds. These species contain two hydroxyl (boronic acids) or two alkoxy (boronic esters) groups along with one organic group. Boronamides contain two nitrogen atoms bonded to boron.
Boronic acidA boronic acid is a compound related to in which one of the three is replaced by an or group. As a compound containing a, members of this class thus belong to the larger class of. Boronic acids act as. Their unique feature is that they are capable of forming reversible covalent complexes with, etc.
(molecules with, (1,2) or occasionally (1,3) substituted Lewis base donors (, carboxylate)). The of a boronic acid is 9, but they can form boronate complexes with p K a 7. They are occasionally used in the area of molecular recognition to bind to saccharides for fluorescent detection or selective transport of across membranes. Boronic acids are used extensively in as chemical building blocks and intermediates predominantly in the.
A key concept in its chemistry is of its organic residue to a transition metal.The compound with a boronic acid group is a drug used in. The boron atom in this molecule is a key substructure because through it certain are blocked that would otherwise degrade proteins.
Boronic acids are known to bind to active site serines and are part of inhibitors for porcine, and the protease. Furthermore, boronic acid derivatives constitute a class of inhibitors for human 1 and 2, which are cancer drug targets within the cycle.The boronic acid functional group is reputed to have low inherent toxicity. This is one of the reasons for the popularity of the Suzuki coupling in the development and synthesis of pharmaceutical agents. However, a significant fraction of commonly used boronic acids and their derivatives were recently found to gives a positive Ames test and act as chemical. The mechanism of mutagenicity is thought to involve the generation of organic radicals via oxidation of the boronic acid by atmospheric oxygen. The general structure of a boronic acid, where R is a.
Structure and synthesisIn 1860, was the first to report the preparation and isolation of a boronic acid. Ethylboronic acid was synthesized by a two-stage process. First, and reacted to produce. This compound then in air to form ethylboronic acid. Several synthetic routes are now in common use, and many air-stable boronic acids are commercially available.Boronic acids typically have high melting points. They are prone to forming by loss of water molecules, typically to give cyclic.Examples of boronic acidsBoronic acidRStructure°C121.93–2192-Thienylboronic acid127.9661–140Methylboronic acid-96-691–94cis-Propenylboronic acid85.907547-96-865–70trans-Propenylboronic acid85.9075–127SynthesisBoronic acids can be obtained via several methods.
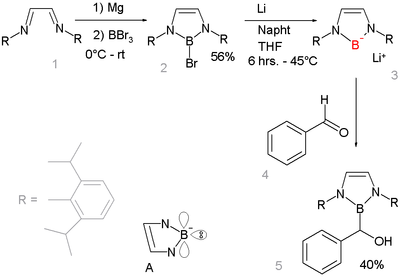
The most common way is reaction of organometallic compounds based on lithium or magnesium with. For example, is produced from and followed by hydrolysis PhMgBr + B(OMe) 3 → PhB(OMe) 2 + MeOMgBr PhB(OMe) 2 + H 2O → PhB(OH) 2 + MeOHAnother method is reaction of an (RSiR 3) with (BBr 3) in a to RBBr 2 followed by acidic hydrolysis.A third method is by palladium catalysed reaction of aryl halides and triflates with diboronyl esters in a. An alternative to esters in this method is the use of diboronic acid or tetrahydroxydiboron (B(OH 2) 2). Boronic esters (also named boronate esters)Boronic esters are formed between a boronic acid and an alcohol. The sequence is of the amine, of the amine to the copper(II), (transferring the alkyl boron group to copper and the copper acetate group to boron), of Cu(II) to Cu(III) by oxygen and finally of Cu(III) to Cu(I) with formation of the product.
Direct reductive elimination of Cu(II) to Cu(0) also takes place but is very slow. In oxygen also regenerates the Cu(II) catalyst.Liebeskind–Srogl couplingIn the a is coupled with a boronic acid to produce a.Conjugate additionThe boronic acid organic residue is a nucleophile in also in conjunction with a metal. In one study the pinacol ester of allylboronic acid is reacted with in such a conjugate addition.
Homologization applicationIn this reaction converts the boronic ester into a boronate. A then induces a rearrangement of the alkyl group with of the chlorine group.
Finally an such as a displaces the second chlorine atom effectively leading to insertion of an RCH 2 group into the C-B bond. Another reaction featuring a boronate alkyl migration is the.Electrophilic allyl shiftsAllyl boronic esters engage in very much like silicon pendant in the.
In one study a diallylation reagent combines both. Protonolysisis a chemical reaction involving the of a boronic acid (or other organoborane compound) in which a carbon-boron bond is broken and replaced with a carbon-hydrogen bond. Protodeboronation is a well-known undesired, and frequently associated with metal-catalysed that utilise boronic acids (see ). For a given boronic acid, the propensity to undergo protodeboronation is highly variable and dependent on various factors, such as the reaction conditions employed and the organic substituent of the boronic acid.
