Highly pure proteins are vital for successful experiments; they play roles in research as assay reagents (especially for ), therapeutic candidates, and of course, as the subjects of structural and biochemical studies. Is the science of separation and we utilize it to isolate and purify proteins based on their unique physiochemical properties. One of the most fundamental and important skill sets a budding life scientist can master is protein chromatography.This is the first in a series of articles written to introduce the various separation methods and provide some practical tips. We will begin with ion-exchange chromatography.
A Brief Overview of Ion-Exchange Chromatography(IEX) separates biomolecules based on differences in their net charge at a particular pH. Protein charge depends on the number and type of ionizable amino acid side chain groups. Each protein has an isoelectric point (pI), a pH at which the overall number of negative and positive charges is zero. In a buffered solution below the protein’s pI, the protein is positively charged and will bind to the negatively charged functional groups of a cation exchange resin. In a buffered solution above the protein’s pI, the protein is negatively charged and will bind to the positively charged functional groups of an anion exchange resin. In principle, a protein could bind to either a cation or anion exchange resin, but in practice, proteins are only stable within a narrow pH range and the choice of resin depends on the pH stability of the protein.
A complete family of columns for inorganic cations and amines. You'll get high-resolution separation of inorganic cations, ammonium, and amines using these Thermo Scientific™ Dionex™ IonPac™ cation-exchange columns. They are excellent for separations of alkali metals, alkaline earth metals, alkylamines, alkanolamines and biogenic amines. However, one area that has lagged in chromatography development is cation exchange chromatography (CEX) for charge variant analysis. Polyether ether ketone (PEEK) is the most common column hardware for CEX, however, variations in column inner diameter and low pressure limitations render it incapable of consistent UHPLC applications.
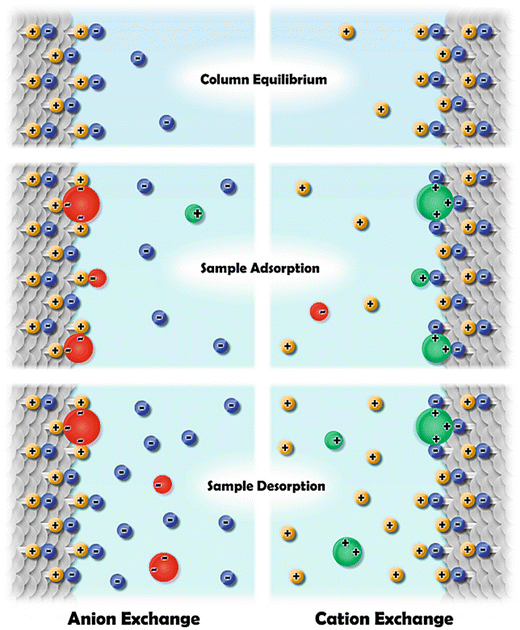
Resins for Ion-Exchange ChromatographyIon-exchange chromatography resins have charged functional groups bound to resin beads which attract biomolecules of the opposite charge. Cation exchange resins are negatively charged, and anion exchange resins are positively charged.
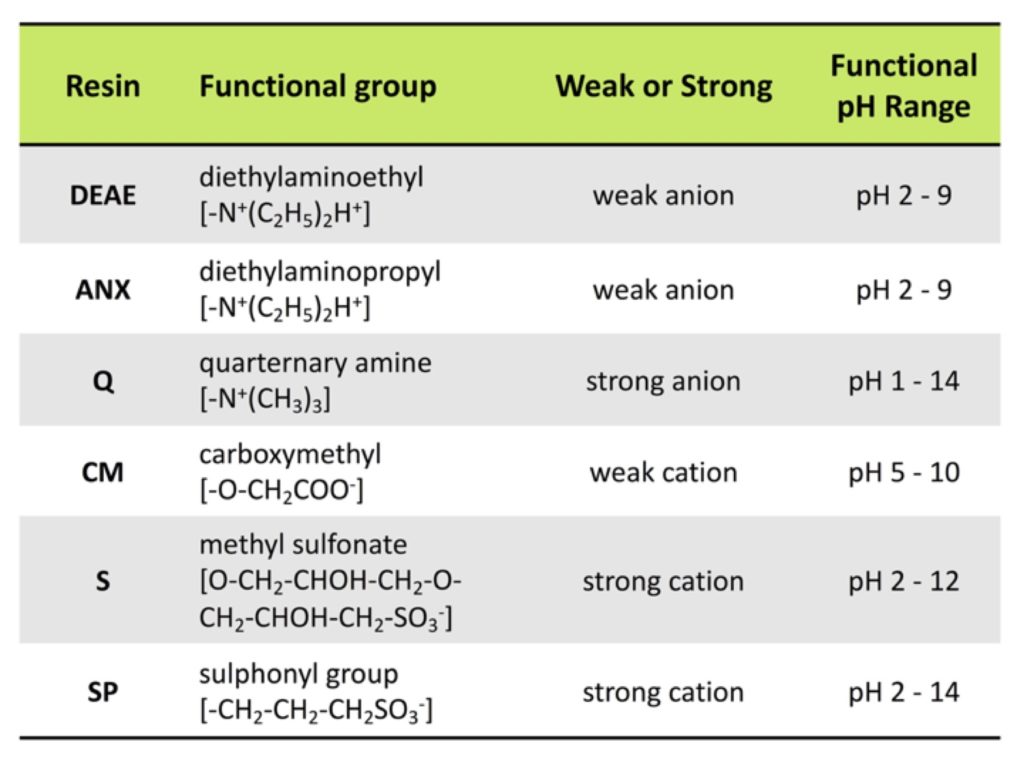
Ion-exchange resins are also categorized as “weak” or “strong” exchangers. These terms aren’t related to the strength of ion binding, but instead, refer to the extent that the ionization state of the functional groups varies with pH. A “weak” exchanger is ionized over only a limited pH range, while a “strong” exchanger shows no variation in ion exchange capacity with changes in pH. Weak exchange resins can take up or lose protons with changes in buffer pH, and that added variation in charge offers an additional dimension of selectivity for binding and elution. Strong exchangers do not vary and remain fully charged over a broad pH range, which can make optimization of separation simpler than with weak exchangers. The table below summarizes the most common IEX resins.In addition to type of functional group, you need to consider the physical properties of the resin.
The size, material and porosity of the resin beads dictate the maximum operating pressures and flow-rates (affects the speed of purification). More importantly, bead size and porosity affect the resolution of the separation. Larger beads are generally conducive to fast flow rates and provide resolution appropriate for early and intermediate stages of purification.
Smaller beads provide the best resolution and are ideal for later purification stages. Table 1: Summary of ion-exchange chromatography resins & their functional groups. Easy as pIThe first step in designing an IEX purification scheme could be the in silico determination of the pI of your protein of interest. The pI of a protein is determined by the aggregate charge of every amino acid in the protein chain.
The Henderson-Hasselbach equation is used to iteratively compute protein charge at certain pH’s until one is found in which the net charge of the protein is zero. The math can get complicated for proteins, but luckily, there are several online tools you can use to painlessly estimate your protein’s pI and guide your experiments. This is the “classic” tool that most life scientists know of.
You can calculate the theoretical molecular weight, isoelectric point, extinction coefficient and other physiochemical properties based on protein sequence. The algorithm is based off the work of Bjellqvist et al. In the early 1990’s –.
The IPC is a “new school” online tool. It computes a series of pI predictions using several published algorithms and pK datasets. In addition to displaying the range of computed isoelectric points, it also provides an average pI based for all methods. I find the output from IPC to be more useful and comprehensive as compared to the value determined by ProtParam. The. Post-translational modifications (PTM) can have a significant effect on a protein’s pI. The majority of PTMs occur on ionizable sidechains, and some PTMs, such as phosphorylation or acetylation, introduce new ionizable chemical groups.
If you are purifying a eukaryotic protein that is likely post-translationally modified, use the ProMoST tool to calculate and compares the effect of post-translational modifications on pI, molecular mass, and other physiochemical properties.Remember that the theoretical pI is likely to be different from the true isoelectric point, and may not reflect the actual charge distribution on the protein surface. Charge distribution is not uniform and a protein is capable of having both positively and negatively charged patches on a surface. When working with a new protein, I always screen a range of cation and anion exchangers, both weak and strong, to optimize the purification step. Buffer ConditionsThe choice of buffer system, pH, additives and salt concentration all have a direct effect on the success of your IEX experiment. Buffer scouting is frequently required to find the optimal pH for solubility and adsorption to the IEX resin.
When screening resins and buffer conditions, keep the following in mind:. Keep the pH of any protein purification or storage buffer 0.5 to 1 pH units above or below its pI to promote solubility. A protein’s pI is the point at which it has no net charge; it is likely to be unstable, less reactive, and least soluble at that pH. It’s a simple concept, but may not be the first thing that comes to mind if your protein begins crashing out of solution during buffer exchange. Use a buffer concentration sufficient to maintain buffering capacity, typically 25 to 100 mM.
Pay attention to the ionic strength of the starting material and wash buffers, as the affinity of the protein for the column decreases as ionic strength increases due to salt concentration.
